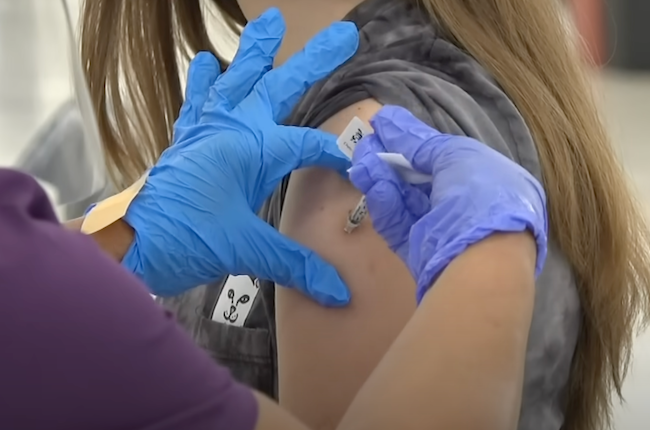
COVID exposed how laws around medicine can prioritise profit over patients — is it time to rethink access to medicine as a human right? Gerry Georgatos reports.
THE COVID-19 PANDEMIC reawakened longstanding tensions between intellectual property protection and public health imperatives.
While patents play a vital role in incentivising pharmaceutical innovation, they can simultaneously act as barriers to access, especially during global health crises.
This tension has prompted renewed scrutiny of the Australian patent system, particularly its breadth and flexibility in responding to emergencies. The argument that Australian patent law is too broad – particularly considering pricing controversies and access limitations during pandemics – demands careful evaluation. I have attempted to examine the statutory and judicial framework of Australian patent law, explore its application in pharmaceutical contexts, analyse the practical consequences for healthcare access and consider reform proposals to ensure a more balanced and responsive legal framework.
Australia has the benefaction of the Pharmaceuticals Benefits Scheme (PBS), which caps listed medications at affordable prices. Australia’s PBS is one of the world's leading prescribed medications provision programs. However, due to exclusivity strategies and prohibitive patents, many medications, particularly newly discovered ones, can be unaffordable.
Despite the argument that patents drive innovation and competition, affordable access at the individual level while a medication is in waiting to be listed with the PBS or delayed by patent strategies, can be unaffordable for many desperate patients.
Balancing innovation and access is a perennial struggle. Evaluating the breadth of Australian patent law amidst pandemics and pharmaceutical pricing controversies is an eye-opener to the monetisation of pharmaceutical healthcare in general, regarding research and discovery as property, against the public good reckoning.
Australian patent law is grounded in the Patents Act 1990, which provides the statutory regime for granting, enforcing, and limiting patent rights. Under section 18(1), an invention must be a manner of manufacture, novel, involve an inventive step, and be useful to qualify for patent protection. These criteria are deliberately broad and have been interpreted expansively by the courts.
The High Court in National Research Development Corporation v Commissioner of Patents established a liberal standard for what constitutes a patentable invention, laying the foundation for the protection of a wide range of technologies, including pharmaceuticals. The decision in D'Arcy v Myriad Genetics reaffirmed that while the courts are willing to constrain patentability in exceptional cases – such as isolated genetic material – they generally uphold a robust scope of protection.
Subsequent cases have further entrenched this breadth. In Grant v Commissioner of Patents, the court endorsed a liberal interpretation of "manner of manufacture," although it drew the line at business methods with no physical aspect. In AstraZeneca AB v Apotex, patent protection was granted for a new dosage regime, raising concerns about evergreening. In pharmaceutical terms, evergreening refers to strategies used by drug companies to extend the life of a patent beyond its original expiration date, thereby delaying the entry of generic competitors into the market. This practice is often criticised to maintain monopoly pricing and restrict access to affordable medicines, especially in the context of public health needs.
Common evergreening tactics include minor modifications to the original drug, such as new dosage forms (for example, extended-release versions), slight chemical alterations (for example, new salt forms or isomers), new methods of administration (such as inhaler vs pill). Furthermore, there is the ascent to patient thickets, the filing of multiple overlapping patents on various aspects of a drug (composition, use, manufacturing process and so on), making it harder for generics to enter the market without risk of litigation.
There is also the leveraging of regulatory protections to delay approval of generics even after patent expiry. Secondary patents are also possible, the filing of patents for new uses of an already known drug (also called “indication patents”). A company holds a patent for Drug A, which is about to expire. Before expiration, it develops Drug A-ER (extended-release) and patents it as a “new invention,” thus gaining additional years of patent protection and delaying generic competition.
Evergreening is seen by many as anti-competitive and contrary to the goals of patent law, which arguably are to incentivise genuine innovation while eventually allowing public access to affordable versions. Evergreening can inflate healthcare costs, particularly affecting developing countries and vulnerable populations. In any nation, citizens in the lowest quintiles of income-base are impacted disproportionately in terms of affordability.
In countries like Australia, evergreening has been a contentious issue. The PBS and legislation such as the Therapeutic Goods Act 1989 interact with the Patents Act 1990 in ways that can either enable or limit evergreening. Legal reforms have sought to curb abuse, for example through anti-evergreening provisions introduced after the Australia–U.S. Free Trade Agreement (AUSFTA).
Similarly, in H Lundbeck A/S v Alphapharm, the Federal Court upheld a controversial patent extension, allowing monopoly pricing to persist despite generic readiness. This broad framework is reinforced by Australia's international obligations. As a signatory to the Agreement on Trade-Related Aspects of Intellectual Property Rights (TRIPS), Australia must comply with minimum standards of patent protection.
Article 27 mandates that patents be available for any inventions, in all fields of technology, provided they are new, involve an inventive step and are capable of industrial application. While TRIPS allows for exceptions, including measures to protect public health, the scope of domestic discretion is limited. The Doha Declaration on TRIPS and Public Health (2001) reaffirmed the right of countries to use TRIPS flexibilities to ensure access to medicines. However, Australia’s implementation of these flexibilities has been conservative and sporadic.
One of the key issues with the breadth of Australian patent law is its impact on pharmaceutical pricing. During the COVID-19 pandemic, patent rights held by pharmaceutical companies over vaccines and treatments became a focal point of global debate. Although companies cited the high cost of research and development to justify exclusive rights, public health advocates criticised this stance for perpetuating inequality in vaccine distribution. As Sampat and Shadlen argue, reliance on patents during a pandemic entrenches systemic barriers and favours high-income countries. Australia, though not a manufacturing hub, was exposed to delayed access and limited bargaining power.
Another key episode involved the hepatitis C drug Sofosbuvir, marketed as Sovaldi. Initially priced at $84,000 for a 12-week course, the drug threatened the sustainability of the PBS. While subsequent negotiations reduced the cost, the episode highlighted how patent monopolies, once granted, can create prolonged access barriers even in publicly subsidised systems.
Australian patent law includes mechanisms intended to address these concerns, but their effectiveness is questionable. Section 133 of the Patents Act permits compulsory licensing if reasonable requirements of the public are not being met. Yet, despite multiple public health controversies, Australia has never granted a compulsory licence. The case of Apple v Samsung Electronics involved attempted use of compulsory licensing, but the Federal Court set a high bar for granting such orders, focusing on commercial rather than public health criteria.
The Crown use provisions, located in sections 163 to 170, allow the government to use patents in the public interest. Amended in 2020, these provisions are now somewhat easier to invoke, yet no significant public health intervention has occurred under them. Even during COVID-19, Australia did not exercise Crown use powers for vaccines or treatments, in contrast to global calls for open licensing.
Furthermore, provisions in the Therapeutic Goods Act and the National Health Act 1953 regulate market entry and reimbursement. However, patent-related delays often affect listings on the PBS, compounding affordability issues. Patent linkage, although not formalised as in the U.S., exerts a chilling effect on generic entry due to litigation threats.
Patent linkage in pharmaceuticals refers to a legal or regulatory system that links the approval of generic drugs to the patent status of the original (brand-name) drug. Under such a system, regulatory authorities (like the Therapeutic Goods Administration in Australia or the FDA in the U.S.) cannot approve a generic drug for market entry until they have considered the patent rights of the originator drug.
The generic manufacturer may be required to notify the patent holder if it seeks approval before the expiry of a patent. The regulatory body may be required to delay the approval of a generic if the patent holder initiates legal action within a specific period. A public register may list patents associated with approved drugs, helping regulators and generic companies determine which patents might be infringed by a new generic. The stated goal is to prevent patent infringement by ensuring that generics do not enter the market while a valid patent is still in force. In practice, the grim reality is that it often serves to delay the market entry of generics, potentially contributing to evergreening.
Critics argue that patent linkage gives excessive power to originator companies, enabling them to delay generic competition through litigation or frivolous patent listings. It can be used strategically, rather than to protect genuine innovation, leading to higher drug costs and restricted access.
Australia does not have a full patent linkage system like the U.S. Hatch-Waxman Act, but elements of it were introduced as part of the AUSFTA. The Therapeutic Goods Act requires generic applicants to certify that they will not infringe any valid patents, or that they have notified the patent holder of their intent to market before patent expiry. Australia added anti-evergreening provisions to counterbalance the AUSFTA-linked changes, such as penalties for false or misleading patent enforcement claims.
The pharmaceutical industry maintains that robust patent rights are necessary to fund high-risk innovation. This is reflected in Bristol-Myers Squibb Company v Apotex, where the court upheld patent rights over minor chemical modifications. Economic analyses support this incentive model, but critics like Faunce and Townsend argue that monopoly-based pricing distorts access priorities. The practice of evergreening – pursuing additional patents for minor modifications – has received criticism, particularly following the Federal Court’s ruling in Apotex v& Sanofi-Aventis Australia, which permitted new-use patents despite lacking substantial therapeutic innovation.
Other jurisdictions demonstrate alternative approaches. India's section 3(d) of the Patents Act prevents patents for new forms of known substances unless they demonstrate enhanced efficacy. This allowed India to deny Novartis a patent for Glivec, preserving generic competition. Australia lacks any comparable statutory limitation.
Even the USA has contemplated limits through "march-in rights" under the Bayh-Dole Act, though unused to date. Europe’s Supplementary Protection Certificates (SPCs) have also faced criticism yet are constrained by clearer conditions compared to Australia’s flexible extensions of term under section 70 of the Patents Act.
Scholars such as Weatherall and Rimmer advocate reforms to prevent abuse of the system while preserving genuine innovation incentives. Proposals include establishing an independent review panel for Crown use applications, capping term extensions, and enacting a public interest override mechanism. Others argue for tightening the definition of “inventive step,” referencing Genentech Inc v Commissioner of Patents, where a low threshold for obviousness enabled protection of incremental innovations.
The Productivity Commission's 2016 Intellectual Property Arrangements Inquiry recommended narrowing the scope of patentable subject matter and reforming the Crown use and compulsory licensing provisions. While some changes followed, core recommendations – such as excluding software and business methods or reducing evergreening – remain unimplemented.
The Productivity Commission's 2016 Intellectual Property Arrangements Inquiry Report offered a comprehensive critique of Australia's intellectual property (IP) system, particularly in relation to the patent system's scope, effectiveness, and public interest alignment. The Commission identified several inefficiencies and anti-competitive practices – most notably in pharmaceuticals – and made several core recommendations, many of which remain partially or wholly unimplemented.
The Commission argued that Australia’s broad interpretation of what constitutes patentable subject matter – particularly under section 18 of the Patents Act – has led to the granting of patents for low-threshold or incremental innovations, including software, business methods, and evergreening pharmaceutical modifications. This was seen as inconsistent with the objectives of the patent system: to promote genuine innovation and disseminate knowledge, while avoiding monopolies that unduly restrict access.
Core recommendations included tightening the "manner of manufacture" test. The Inquiry deliberated the importance of replacing or clarifying the outdated “manner of manufacture” criterion with clear legislative language defining what is patentable, especially for emerging and digital technologies. Exclude certain subject matter from patentability, including software, where the inventive step lies only in the source code or abstract logic. The Inquiry recommended raising the inventive step threshold: require that an invention not be obvious to a person skilled in the art without creativity or extended effort, aligning Australia more closely with European standards.
The Inquiry challenged evergreening, recommending the avoidance of granting patents for minor or strategic modifications to existing drugs, unless they provide genuine therapeutic advances. Another recommendation argued tightening the utility and disclosure requirements to ensure the claimed benefits are real and substantial.
The Inquiry found that compulsory licensing and Crown use mechanisms – which allow governments or third parties to use patented inventions without the patent holder’s consent in the public interest – were underused and inefficient due to legal and procedural complexity.
Summarily, recommendations for reform emphasised simplify and clarify access to compulsory licensing. The threshold for use should not require proving that the patent holder is refusing to license on reasonable terms. Instead, focus on whether access to the patent is in the public interest — especially in situations involving health emergencies, anti-competitive conduct, or market failure.
Merging the Crown use and compulsory licensing regimes by introducing a single, streamlined framework for public interest access to patented technologies. Allow both government and private entities to seek access for public purposes (for example, health, environment, infrastructure). Make it more proactive and transparent and require regular reporting and review mechanisms. Consider upfront pricing guidance or arbitration for disputes over reasonable compensation.
In terms of a public health and pandemic response, ensure the system can support emergency responses, such as allowing generic production of essential medicines or vaccines without excessive delay or litigation risk.
Despite the robust findings, the government made modest reforms, such as minor changes to Crown use in 2020 via the Intellectual Property Laws Amendment (Productivity Commission Response Part 2 and Other Measures) Act 2020. However, key recommendations were not implemented, including the exclusion of software and business methods, nor a higher inventive step, nor stronger anti-evergreening rules and, nor comprehensive reform of compulsory licensing and public interest access.
Recent commentary also supports price transparency reforms in the PBS and greater public-sector involvement in early-stage pharmaceutical research, thereby reducing dependence on monopoly pricing. Open licensing, patent pooling (for example, through the Medicines Patent Pool), and global initiatives like C-TAP (COVID-19 Technology Access Pool) have also been proposed as models for equitable access.
In conclusion, the breadth of Australian patent law, while designed to foster innovation, presents significant challenges in the context of pandemics and pharmaceutical pricing controversies. The current legal framework grants extensive rights to patent holders, often at the expense of timely and affordable access to essential medicines and therefore to the detriment of individuals.
Although mechanisms such as compulsory licensing and Crown use exist, their practical utility is undermined by procedural complexity and political hesitancy. A more balanced approach is needed — one that safeguards innovation incentives while prioritising public health. This requires legislative reform, policy innovation, and a willingness to critically reassess the assumptions underlying the patent system. Only then can Australian patent law truly serve both innovation and access in an equitable manner.
I have argued that while the economic justifications for robust patent protection remain relevant, they cannot be viewed in isolation from public health imperatives. The COVID-19 pandemic and ongoing pricing controversies have made it clear that access to medicines is not merely a policy issue but a fundamental component of human rights.
Australian patent law must evolve to reflect this reality by narrowing overly broad protections, facilitating timely interventions in public emergencies, and embedding public interest considerations more deeply within its legal architecture.
Gerry Georgatos is a suicide prevention and poverty researcher with an experiential focus on social justice.
 This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Australia License
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Australia License
Support independent journalism Subscribe to IA.